Iveric Bio Announces New Functional Vision Loss Reduction Data from Avacincaptad Pegol GATHER Trials

Iveric bio announced new findings from exploratory analyses of data for avacincaptad pegol (ACP), which were presented at the 2023 annual meeting of the Association for Research in Vision and Ophthalmology (ARVO) in New Orleans.
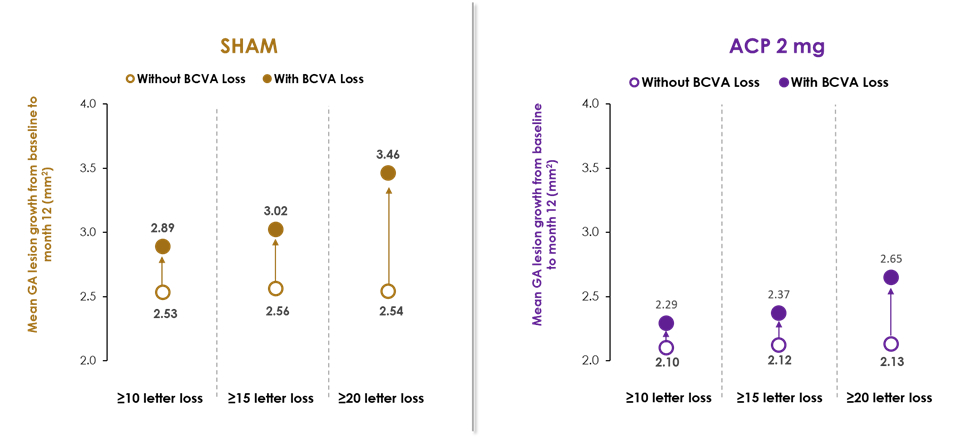
A post-hoc analysis from the GATHER1 and GATHER2 pivotal phase 3 clinical trials showed, for the first time in an interventional study in GA, a relationship between GA growth and worsening vision loss. In this combined analysis, greater vision loss was correlated with increased GA growth.
“This is the first time a relationship between disease progression and worsening visual acuity has been observed in GA, connecting anatomy and function,” Carl Danzig, MD, Director, Vitreo-Retinal Services, Rand Eye Institute, Deerfield Beach, Florida, who presented the findings at ARVO, said in a company news release. “These data suggest that in the ACP-treated group, the reduction in growth of GA resulted in an overall lower rate of vision loss.”
As previously announced, the post-hoc analysis of GATHER1 and GATHER2 combined data signaled a 56% risk reduction in the rate of persistent vision loss in GA patients receiving ACP 2 mg compared to sham over the first 12 months of treatment. Persistent vision loss was defined as a loss of ≥15 letters in Best Corrected Visual Acuity (BCVA) from baseline measured at any two consecutive visits up to month 12. Multiple sensitivity analyses were conducted to evaluate this finding, including 10- and 20-letter loss at two consecutive visits up to month 12, and results were consistent.
Avacincaptad Pegol is currently under evaluation for safety and efficacy by the FDA. A new drug application was granted Priority Review with a Prescription Drug User Fee Act (PDUFA) date of August 19, 2023. The FDA has not identified any potential review issues and the FDA is not currectly planning to hold an Advisory Committee meeting for ACP.
