Iveric Bio Announces Foveal Anatomy Post-Hoc Analysis from GATHER1 Clinical Trial of Zimura in Patients with GA

Iveric bio announced a post-hoc analysis which evaluated various geographic atrophy (GA) growth parameters to explore the rate of disease progression within various regions in the fovea in a subset of patients from the GATHER1 Zimura (avacincaptad pegol) phase 3 clinical trial for the treatment of GA.
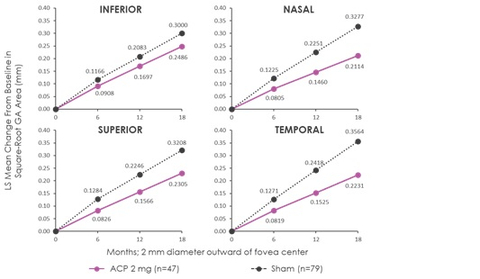
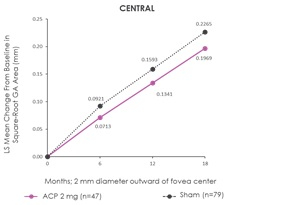
“Consistent with the overall results of GATHER1, in the new analysis a reduction in lesion growth in five standardized regions surrounding and including the central foveal area was observed for patients receiving Zimura 2 mg as compared to patients receiving sham over a period of 18 months,” Dhaval Desai, PharmD, Chief Development Officer of Iveric Bio, said in a company news release. “We believe the observed pattern of reduction in GA growth is consistent with the natural history of the disease and recent clinical trial results in which complement inhibition has been observed to be associated with a greater reduction in GA growth in patients with non-foveal GA, which is known from the natural history to be faster progressing than foveal-involving GA. This analysis supports our expectation that we would see a greater reduction in growth away from the foveal center, reflecting the circumferential growth pattern typical for GA patients.”
“Geographic atrophy has a major impact on functional vision which could alter the quality of life and independence of affected individuals,” Pravin U. Dugel, MD, President of Iveric Bio, said in a company news release. “We believe the results from this exploratory analysis are another step in studying the potential of Zimura to preserve central vision by slowing the progression of GA.”
The results of this subgroup analysis are consistent with the primary analysis results in the intent to treat population. The post-hoc analysis evaluated GA growth in five standardized regions in the retina for patients for whom images were available at relevant time points (n=47 in the Zimura 2 mg group and n=79 in the sham group).The five regions included the central foveal region, consisting of a 2 mm diameter circle around the foveal center point, and four quadrants, temporal, nasal, superior and inferior, in a concentric 8 mm diameter circle around the foveal center point. The accompanying schematic illustrates the five standardized regions.
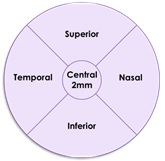
The accompanying graphs illustrate the results of the analysis that will be presented today at the Angiogenesis, Exudation, and Degeneration 2022 meeting by Glenn J. Jaffe, MD, Director, Duke Reading Center Chief, Retina Division, Duke Eye Center, Robert Machemer Professor of Ophthalmology.
About GATHER1 and GATHER2
The company previously announced that GATHER1 showed Zimura (avacincaptad pegol) met its pre-specified primary efficacy endpoint with statistical significance in the phase 3 clinical trial. The most frequently reported ocular adverse events reported with Zimura in this trial were related to the injection procedure. The company expects topline data for GATHER2, a second chase 3 clinical trial for Zimura for GA, to be available in the second half of 2022, approximately 1 year after the enrollment of the last patient in the trial plus the time needed for database lock and analysis. If 12-month results from GATHER2 are positive, the company plans to file applications with the results from GATHER1 and GATHER2 to the FDA and the European Medicines Agency for marketing approval of Zimura for GA. There are no FDA or EMA approved treatments available for patients with GA.
