Genentech Presents Post-Hoc Clinical and Real-World Data for Vabysmo at ASRS
Genentech announced that data from its ophthalmology portfolio will be highlighted in 25 abstracts at the 2023 American Society of Retina Specialists (ASRS) annual meeting, which will be held from July 28-August 1 in Seattle. The data advance the clinical and real-world evidence supporting the use of Vabysmo (faricimab-svoa), the first and only bispecific antibody for the eye, for the treatment of wet age-related macular degeneration (AMD) and diabetic macular edema (DME).
“The clinical and real-world data at ASRS reinforce the improvement in outcomes brought by Vabysmo in two leading causes of vision loss, particularly new analyses suggesting that Vabysmo is associated with less vision-impacting fibrosis than aflibercept,” Levi Garraway, MD, PhD, chief medical officer and head of Global Product Development, said in a company news release. “By improving disease control while offering a potentially less frequent treatment regimen, Vabysmo may help people spend less time managing their condition.”
Vabysmo is currently approved in over 70 countries to treat wet AMD and DME, with more than 1 million doses distributed globally.
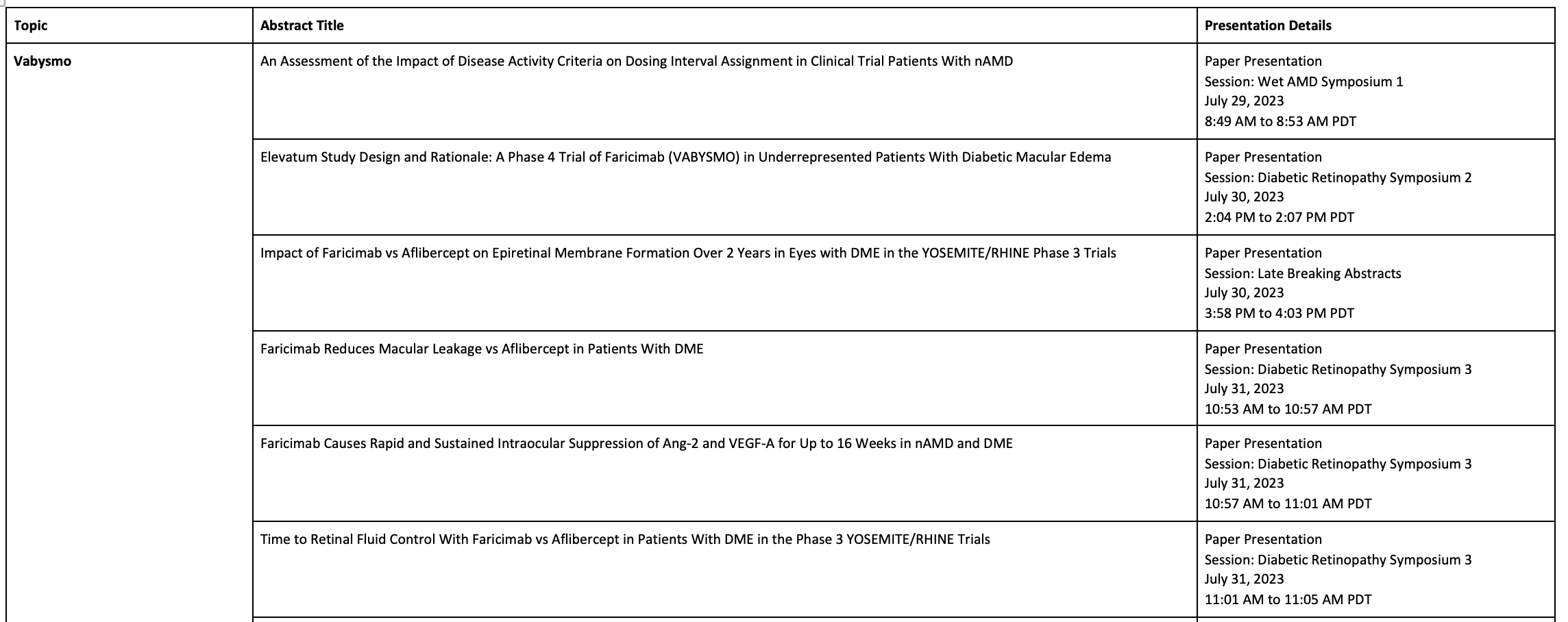
The following data will be presented at ASRS 2023:
Late-breaker: Vabysmo’s effect on epiretinal membrane (ERM) formation in DME compared to aflibercept
Two-year post-hoc data from the YOSEMITE and RHINE phase 3 studies will be presented for the first time on ERM formation in DME patients, indicating Vabysmo leads to less retinal fibrosis than aflibercept.
Vabysmo drying and durability data
Data will be presented reiterating positive anatomical outcomes previously seen with Vabysmo treatment, including reduced blood vessel leakage in the macula, and greater and faster retinal fluid control, according to Genentech.
Data will also further support how increased intervals between doses of Vabysmo to treat wet AMD and DME, compared to aflibercept, do not compromise outcomes.
Vabysmo real-world data
Genentech’s program of real-world studies for Vabysmo includes more than 8,500 participants in almost 30 countries.
- Updates will be presented on real-world data from the FARETINA studies of Vabysmo in wet AMD and DME looking at extended dosing intervals and impact on vision, including Vabysmo’s use as a first-line treatment.
- Preliminary data on early outcomes and treatment patterns in the United Kingdom FARWIDE studies of Vabysmo in wet AMD and DME will be shared for the first time.
In addition, independent investigator studies of Vabysmo are expected to be presented. The TRUCKEE study, which focused on real-world outcomes in people with wet AMD across 14 sites in the United States, is scheduled for presentation on July 31 during the Wet AMD Symposium 3 (1:38 PM to 1:44 PM PDT).