FDA Clears CorNeat EverPatch, World's First Non-Degradable, Synthetic Tissue Substitute for Ophthalmic Surgery

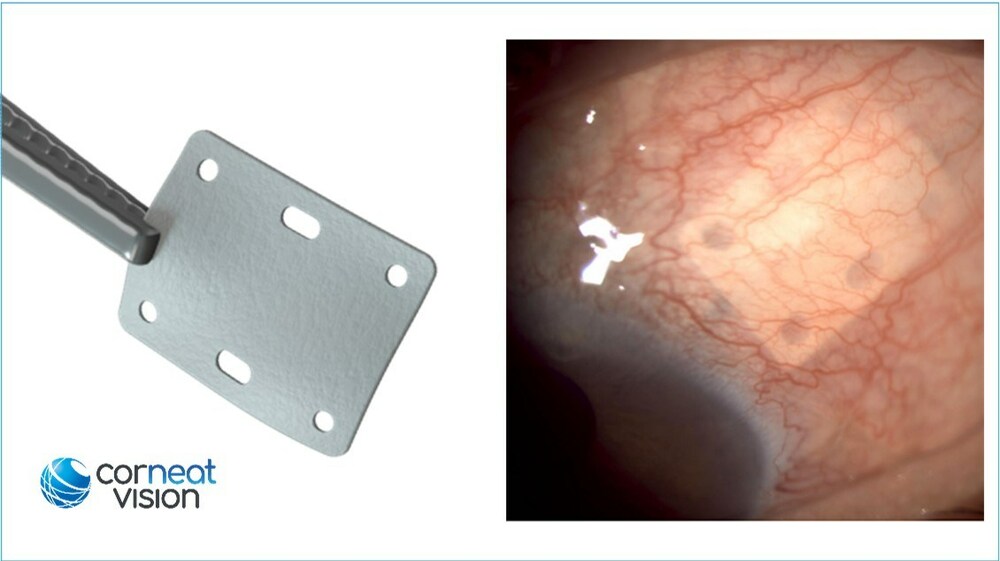
The FDA has granted 510(k) clearance to CorNeat Vision's EverPatch, which the company describes as the first synthetic, non-degradable tissue-integrating matrix for use in ophthalmic surgeries. It is composed of a non-woven, polymer matrix which integrates with surrounding tissue and is intended to reinforce the sclera and aid the physical reconstruction of the ocular surface. This groundbreaking synthetic tissue substitute is designed to address the critical needs of ocular surgeons, providing a sterile and non-degradable solution for patients worldwide.
The CorNeat EverPatch is poised to displace the use of donor and processed tissue, commonly utilized in ocular surgeries but posing the risk of disease transmission.
"The ideal graft material should be long-lasting, sterile, immunologically inactive, cosmetically acceptable, and readily available. The CorNeat EverPatch was designed with these goals in mind," Gilad Litvin, MD, CorNeat Vision's Chief Medical Officer and Co-Founder, said in a company news release. "Our novel ophthalmic patch is significantly thinner than processed patch tissue, provides better handling as it does not 'cheesewire' when sutured, and has holes that allow for accurate positioning and anchoring. These holes also facilitate direct conjunctival adhesion to the sclera thus supporting its bio-integration. Thus far, we have received extremely positive feedback from surgeons and are excited for U.S. surgeons to have this tool available."
"We are extremely excited to introduce the CorNeat EverPatch to the US market. It is the first device that leverages the EverMatrix, our core tissue-integrating material platform technology, originally developed for our corneal prosthesis program," said Almog Aley-Raz, CorNeat Vision's CEO and VP R&D. "EverMatrix presents a significant business opportunity as it is the only synthetic non-degradable patch material in ocular surgery. This biocompatible material has the potential for wider use in soft tissue reinforcement, biomechanical integration of implants with surrounding tissue, fabrication of membranes, and concealment of implants and sensors. I cannot overemphasize the potential impact of this technology on human health. This recent FDA clearance of our first device for ophthalmic use advances us toward our ultimate goal of promoting human health and equality."
The CorNeat EverPatch will be initially launched in select ophthalmic centers in the US in Q3 2023, expanding nationwide later in the year.