FDA Approves Apellis' Syfovre (Pegcetacoplan), First and Only Treatment for Geographic Atrophy

In a historic approval, for the first time patients with geographic atrophy (GA) will have an FDA-approved treatment option. The FDA on Friday approved Apellis Pharmaceuticals' Syfovre (pegcetacoplan) for the treatment of geographic atrophy (GA) secondary to age-related macular degeneration (AMD).
"It was just a great relief to hear that there's finally the first approved treatment for this condition," Eleonora Lad, MD, PhD, lead investigator for the OAKS study, said in an interview with Eyewire News. "This was a big day for all of us. For GA patients, for physicians that treat the patients, families, and everybody in the space that was interested in this news."
Syfovre is expected to be available in the US by the beginning of March through specialty distributors and specialty pharmacies nationwide. It will cost $2,190 per vial before discounts, according to Apellis.
“Today marks an extraordinary milestone for patients, the retina community, and Apellis. With its increasing effects over time and flexible dosing, we believe that Syfovre will make a meaningful difference in the lives of people with GA,” Cedric Francois, MD, PhD, co-founder and chief executive officer, Apellis, said in the news release. “GA is a complex disease that the field has spent decades trying to address, so we are humbled and proud to bring forward the first-ever treatment. Thank you to all who helped make this moment a reality.”
The approval of Syfovre is based on positive results from the phase 3 OAKS and DERBY studies at 24 months across a broad and representative population of patients. Syfovre is approved for GA patients with or without subfoveal involvement and provides dosing flexibility for patients and physicians with a dosing regimen of every 25 to 60 days. In the OAKS and DERBY studies, Syfovre reduced the rate of GA lesion growth compared to sham and demonstrated increasing treatment effects over time, with the greatest benefit (up to 36% reduction in lesion growth with monthly treatment in DERBY) occurring between months 18-24.
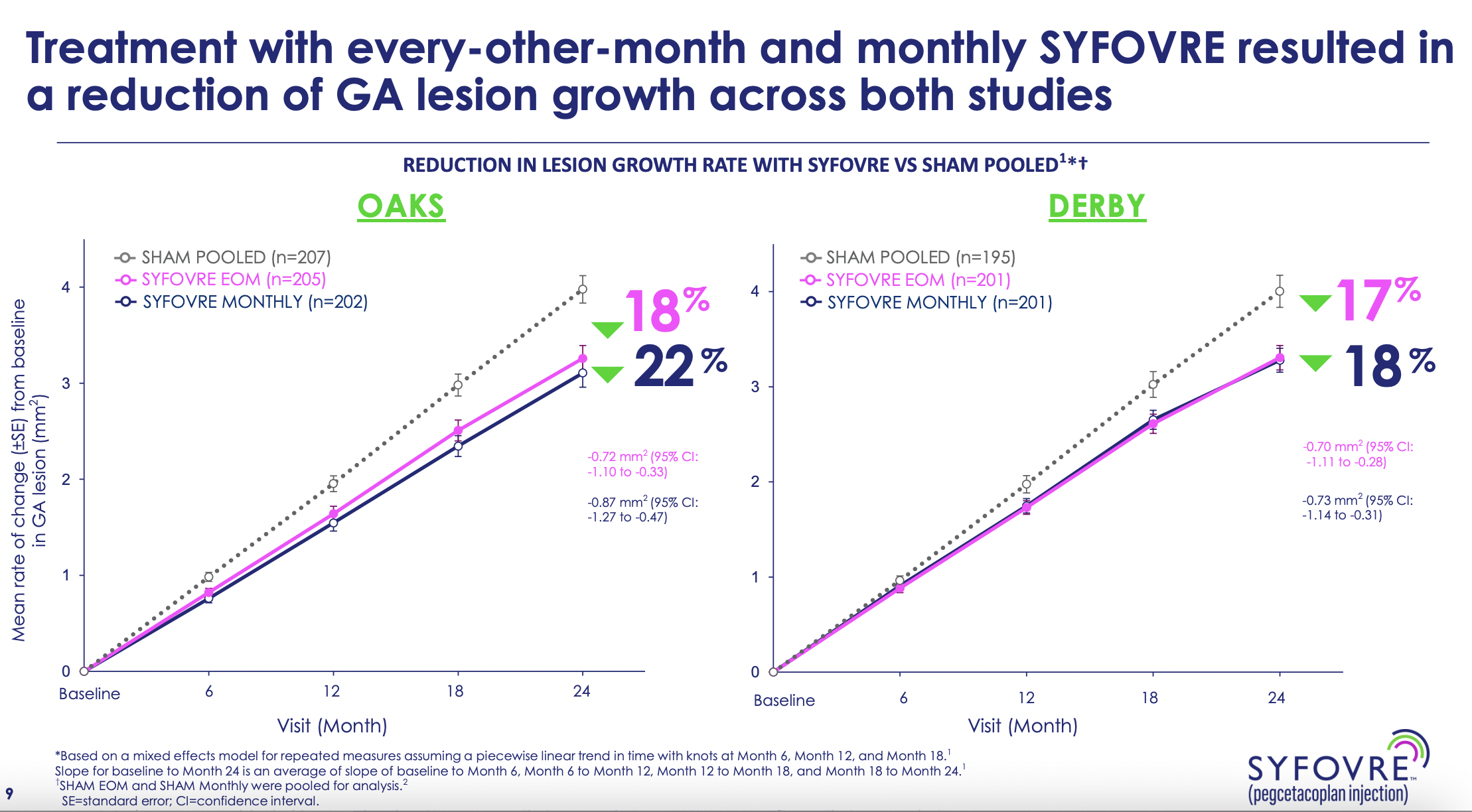
"The most significant finding from the trial and the most impressive in my opinion, was the fact that dosing of this drug over time had increased effects. In other words, the more the patients use this treatment chronically, the more likely they were to be differentiated from the sham group. And that's key in a chronic condition that didn't happen overnight," Dr. Lad told Eyewire News. "So the treatment efficacy accelerate over time, especially between months 18 and 24. This would help us as clinicians guide our patients in the management to explain that they have to stay on treatment for life to achieve maximal benefits in preserving retinal tissue and preserving vision over longer time courses."

The safety profile of Syfovre is well-demonstrated following about 12,000 injections. The most common adverse reactions (≥ 5%) reported in patients receiving Syfovre were ocular discomfort, neovascular AMD, vitreous floaters, and conjunctival hemorrhage. Syfovre is contraindicated in patients with ocular or periocular infections, and in patients with active intraocular inflammation.

Apellis said it is committed to helping patients with treatment access and support. The company set up "ApellisAssist," a program designed to help provide Syfovre patients insurance support, financial assistance for eligible patients, disease education, and ongoing product support. Patients and healthcare providers can call 1-888-273-5547 for more information.
A marketing authorization application for Syfovre is under review by the European Medicines Agency with a decision expected in early 2024. In addition, a marketing application has been submitted to Health Canada.


