CORD LLC Completes One-Year Follow-Up on Subjects From the Model SC9 Intraocular Lens FDA Clinical Trial

Cumming Ophthalmic Research and Development (CORD) LLC announced results from its 1-year follow-up on patients who were implanted with the Model SC9 IOL in a clinical trial. The trial was performed under an investigational device exemption (IDE) with the FDA for the treatment of cataracts. This important milestone enables the company to file a premarket approval (PMA) application to secure FDA approval for the commercial distribution of the Model SC9 in the United States.
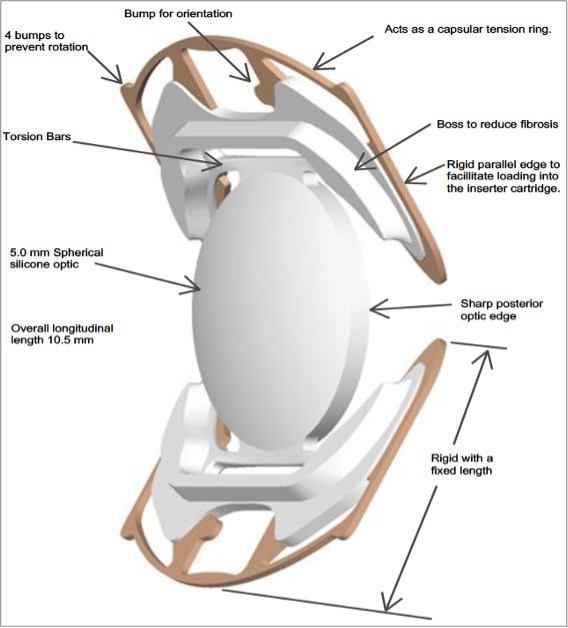
The clinical trial, which included 17 eyes in 15 patients implanted with the Model SC9 lens, was conducted by Juan Batlle, MD, at the Centro de Microcirugia Ocular y Laser in the Dominican Republic. Results from the 1-year follow-up demonstrated:
- Distance corrected near visual acuity (DCNVA) of 20/40 or better in 14 of the 16 eyes available at 12 months (87%)
- Uncorrected near and intermediate visual acuity (UCNVA and UCIVA) of 20/40 or better in 14 of the 16 eyes available at 12 months (87%)
- Postsurgical vision that was better than before the procedure, even with the use of glasses and contact lenses, for all participants
- Uncorrected distance visual acuity – meaning natural vision with no vision aids – was at least 20/40 in all eyes implanted with the SC9 lens
"Dr. Cumming has done it again," said John Hovanesian of Harvard Eye Associates in Laguna Hills, California. "As one of the leading pioneers of IOL development, with the Model SC9 he's showing that he has even more to offer the field of ophthalmology. This new lens offers patients a range of vision with a perfectly focused image."
