CORD Files for Premarket Approval with the FDA for the SC9 IOL for Cataract Surgery

Cumming Ophthalmic Research and Development (CORD) announced that it has submitted a premarket approval (PMA) application to the FDA for the Model SC9 IOL. The SC9 is the brainchild of Stuart Cumming, MD, FACS, FRCOphth, who has over one hundred patents related to IOLs. Dr. Cumming designed the Crystalens, approved by the FDA in 2003.
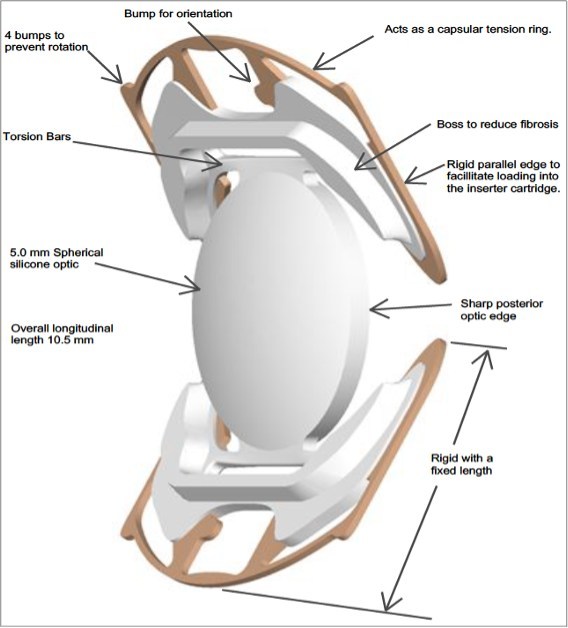
The SC9 lens is designed to treat cataract patients with a single focus spherical optic and a rigid structure to consistently locate the optic in a position intended to provide intermediate vision that is superior to that of a standard monofocal IOL.
CORD is seeking market approval of the SC9 IOL based on the results of more than 300 patients who were followed for a minimum of 1 year under an approved investigational device exemption (IDE).