Apellis' Pegcetacoplan Showed Continuous Effects at Month 18 in Phase 3 DERBY and OAKS Studies for Geographic Atrophy (GA)

Apellis Pharmaceuticals announced longer-term data from the phase 3 DERBY and OAKS studies, which showed that intravitreal pegcetacoplan, an investigational, targeted C3 therapy, continued to reduce geographic atrophy (GA) lesion growth and demonstrate a favorable safety profile at month 18 for the treatment of GA secondary to age-related macular degeneration (AMD). These data will be included in the new drug application (NDA) that the company plans to submit to the FDA in the second quarter of 2022.
“It is exciting to see these positive data with pegcetacoplan, which showed continuous and potentially improving effects over time. These 18-month results provide further evidence that pegcetacoplan meaningfully slows disease progression and has the potential to preserve vision longer,” Jeffrey S. Heier, MD, principal investigator of the DERBY study and director, retina service and director, retinal research, Ophthalmic Consultants of Boston, said in a company news release. “In my practice, I have seen how devastating it can be for people living with GA to lose more of their vision year after year. There is an urgent unmet need in GA, and these results reinforce the potential of pegcetacoplan to become the first-ever treatment for patients with this debilitating disease.”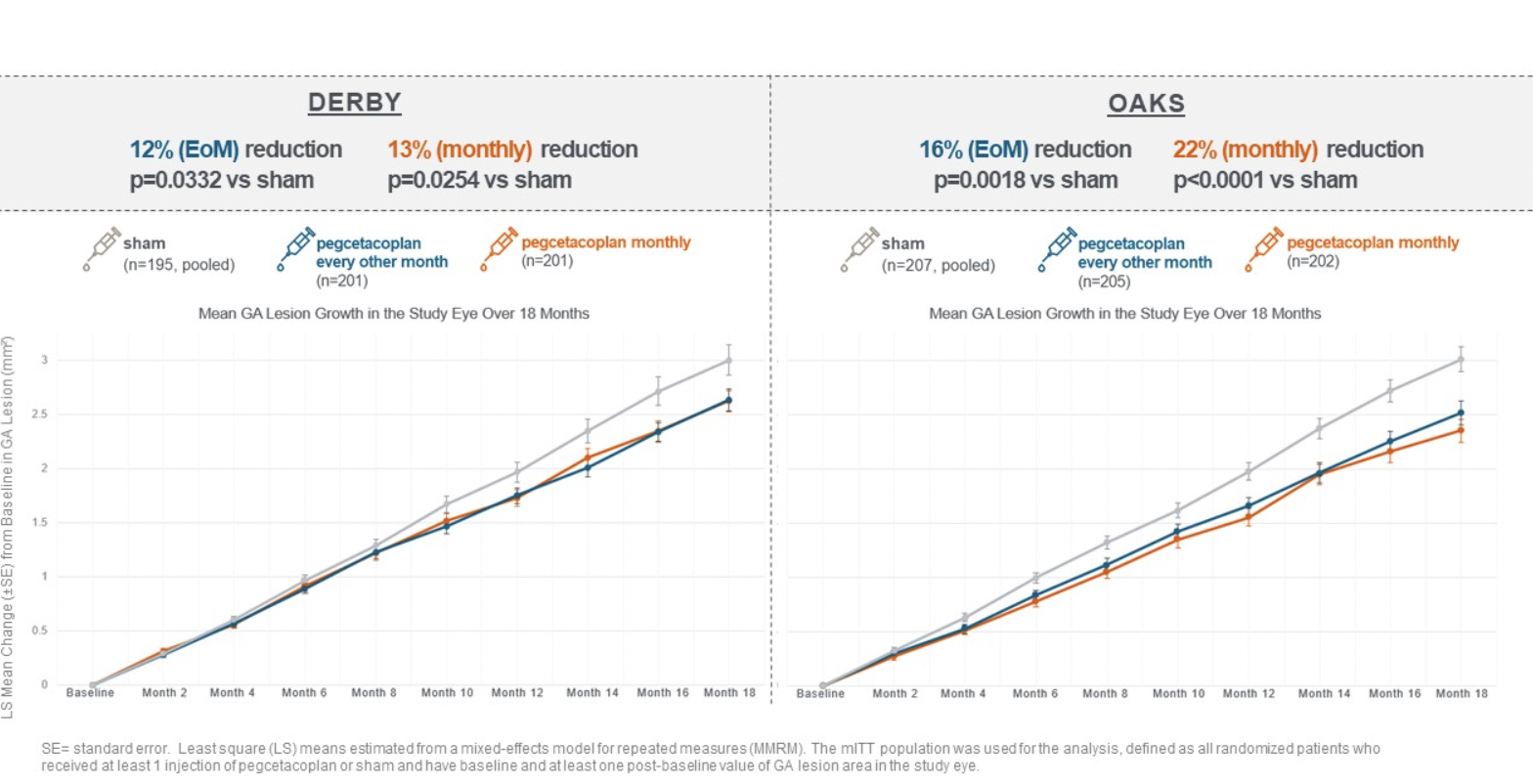
In a longer-term analysis of the primary endpoint, pegcetacoplan continued to reduce GA lesion growth compared to pooled sham at month 18 (all p-values are nominal):
- In OAKS, pegcetacoplan reduced GA lesion growth with both monthly (22%; P<0.0001) and every-other-month treatment (16%; P=0.0018).
- In DERBY, pegcetacoplan reduced GA lesion growth with both monthly (13%; P=0.0254) and every-other-month treatment (12%; P=0.0332).
- Pegcetacoplan demonstrated marked improvements in DERBY during months 6-12 with reductions of 17% with monthly and 16% with every-other-month treatment compared to months 0-6, and the treatment effects were sustained through month 18. The treatment effects observed in DERBY were comparable with OAKS during months 6-18.
- Data at 18 months from the combined studies show the potential for improving treatment effects with pegcetacoplan over time. The reduction in GA lesion growth improved with monthly pegcetacoplan treatment from 13% to 21% from months 0-6 to months 12-18. The reduction in GA lesion growth improved with every-other-month pegcetacoplan treatment from 12% to 17% from months 0-6 to months 12-18.
“Pegcetacoplan is the only therapy to continuously reduce GA lesion growth across a large and broad patient population in phase 3 studies. Building on our 12-month results, both monthly and every-other-month pegcetacoplan demonstrated clinically meaningful effects and a favorable safety profile with longer-term use,” said Federico Grossi, MD, PhD, chief medical officer at Apellis. “We look forward to submitting these data to the FDA and working with regulatory agencies to bring pegcetacoplan to people living with GA around the world as quickly as possible.”
At month 18, pegcetacoplan continued to demonstrate a favorable safety profile, consistent with safety at 12 months and longer-term exposure to intravitreal injections. The rate of infectious endophthalmitis was 0.044% per injection, and the rate of intraocular inflammation was 0.23% per injection. Rates of endophthalmitis and intraocular inflammation continue to be generally in line with those reported in studies of other intravitreal therapies.1,2,3 No events of retinal vasculitis or retinal vein occlusion were observed. The combined rate of new-onset exudations at month 18 was 9.3%, 6.2%, and 2.9% in the pegcetacoplan monthly, every-other-month, and sham groups, respectively.
Detailed data will be presented at upcoming scientific meetings.
References
1Morioka et al. Incidence of endophthalmitis after intravitreal injection of an anti-VEGF agent with or without topical antibiotics. Scientific Reports 2020.
2Kiss et al. Endophthalmitis rates among patients receiving intravitreal anti-VEGF injections: a USA claims analysis. Clin Ophthalmol 2018.
3Cox et al. Inflammatory complications of intravitreal anti-VEGF injections. Journal of Clinical Medicine 2021.
4 Rudnicka AR, Jarrar Z, Wormald R, et al. Age and gender variations in age-related macular degeneration prevalence in populations of European ancestry: a meta analysis. Ophthalmology 2012;119:571–580.
5Wong WL, Su X, Li X, et al. Global prevalence of age-related macular degeneration and disease burden projection for 2020 and 2040: a systematic review and meta-analysis. Lancet Glob Health 2014;2:e106–116.
6 Lindblad AS, et al, and AREDS Research Group. Arch Ophthalmol. 2009;127(9):1168-1174.
7 Seddon, JM, Rosner, B. Validated prediction models for macular degeneration progression and predictors of visual acuity loss identify high-risk individuals. Am J Ophthalmol 2019;198:223–261.

