Aflibercept 8 mg Meets Primary Endpoints in Two Global Pivotal Trials for DME and Wet AMD for 12- and 16-Week Dosing

Regeneron Pharmaceuticals announced that the primary endpoints were met in two pivotal trials investigating novel aflibercept 8 mg with 12- and 16-week dosing regimens in patients with diabetic macular edema (DME) and wet age-related macular degeneration (AMD). The PHOTON trial in DME and the PULSAR trial in wet AMD both demonstrated that aflibercept 8 mg 12- and 16-week dosing regimens achieved noninferiority in vision gains compared to the Eylea 8-week dosing regimen. In these trials, the safety of aflibercept 8 mg was consistent with the established safety profile of Eylea. Regeneron and Bayer will submit these data to regulatory authorities in countries around the world.
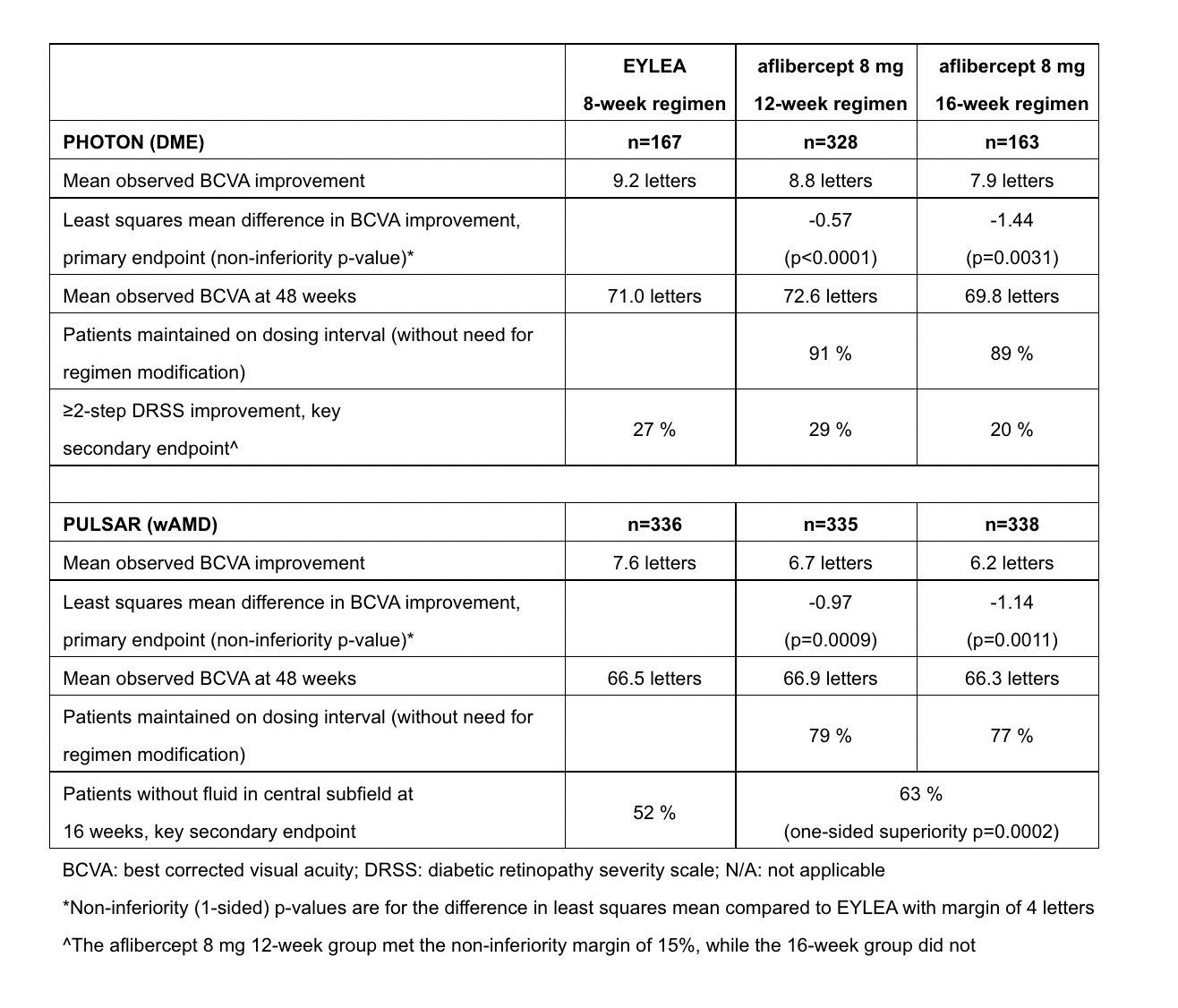
"These pivotal aflibercept 8 mg trials demonstrated that nearly 90% of patients with diabetic macular edema and almost 80% of patients with wet age-related macular degeneration were able to maintain a 16-week dosing regimen," David Brown, MD, FACS, Director of Research at Retina Consultants of Texas in the US and a trial investigator, said in a company news release. "These unprecedented durability data coupled with a safety profile consistent with that of Eylea support aflibercept 8 mg as a potential new standard-of-care in these diseases."
"These groundbreaking results are excellent news for patients. These outcomes have shown that aflibercept 8 mg not only improved vision with less frequent injections, but also demonstrated a similar safety profile to Eylea," said Jean-François Korobelnik, MD, PhD, Professor of Ophthalmology and Head of the Department of Ophthalmology at University Hospital of Bordeaux in France and a trial investigator.
PHOTON (N=658) and PULSAR (N=1,009) are double-masked, active-controlled pivotal trials that are being conducted in multiple centers globally. At 48 weeks, >90% of patients in all dosing groups in both trials completed the treatment period with efficacy results as follows:
The safety of aflibercept 8 mg was similar to Eylea in both trials, and consistent with the well-established safety profile of Eylea from previous clinical trials. In both trials, there were no new safety signals for aflibercept 8 mg and Eylea, and no cases of retinal vasculitis, occlusive retinitis or endophthalmitis. Key safety results* were as follows: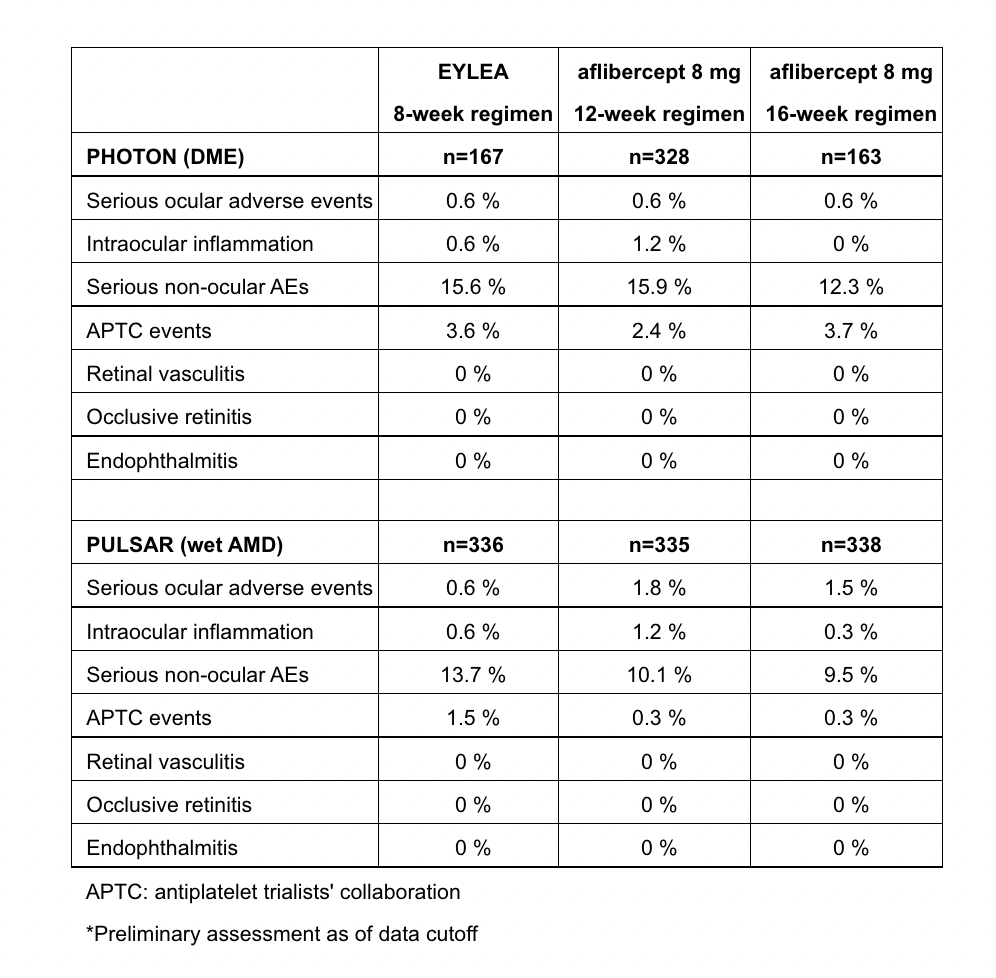
"Over the last decade, Eylea has become the standard-of-care for diabetic macular edema and wet age-related macular degeneration," George D. Yancopoulos, MD, PhD, President and Chief Scientific Officer at Regeneron, and a principal inventor of aflibercept, said in the news release. "The results of these trials with our novel aflibercept 8 mg formulation demonstrated that a remarkably high percentage of patients were maintained on 12- and 16-week dosing intervals through week 48, suggesting aflibercept 8 mg has the potential to be as paradigm-changing as Eylea."
Detailed efficacy and safety data from PHOTON and PULSAR are planned for presentation at an upcoming medical meeting.
Aflibercept 8 mg is being jointly developed by Regeneron and Bayer AG. In the U.S., Regeneron maintains exclusive rights to Eylea and aflibercept 8 mg. Bayer has licensed the exclusive marketing rights outside of the U.S., where the companies share equally the profits from sales of Eylea.
Aflibercept 8 mg is investigational, and its safety and efficacy have not been evaluated by any regulatory authority.
