AcuFocus Announces First Commercial Patients Treated With the IC-8 Apthera IOL

AcuFocus announced that Vance Thompson, MD, of Vance Thompson Vision, Sioux Falls, South Dakota has commercially implanted the first patients with the IC-8 Apthera IOL. The first and only small aperture, non-toric extended depth of focus IOL, the Apthera IOL is FDA approved for the 82% of cataract patients who have as much as 1.5 diopters (D) of corneal astigmatism.[1]
“These initial surgeries mark the start of our controlled limited commercial release of the Apthera IOL,” Al Waterhouse, president and chief executive officer of AcuFocus, said in a company news release. “This is an exciting milestone in our series of firsts: the first small aperture IOL to receive FDA approval, the first lens indicated for implantation with a monofocal or monofocal toric IOL in the fellow eye, the first extended depth of focus lens indicated for monovision, and the first non-toric IOL indicated for cataract patients with low amounts of corneal astigmatism.”
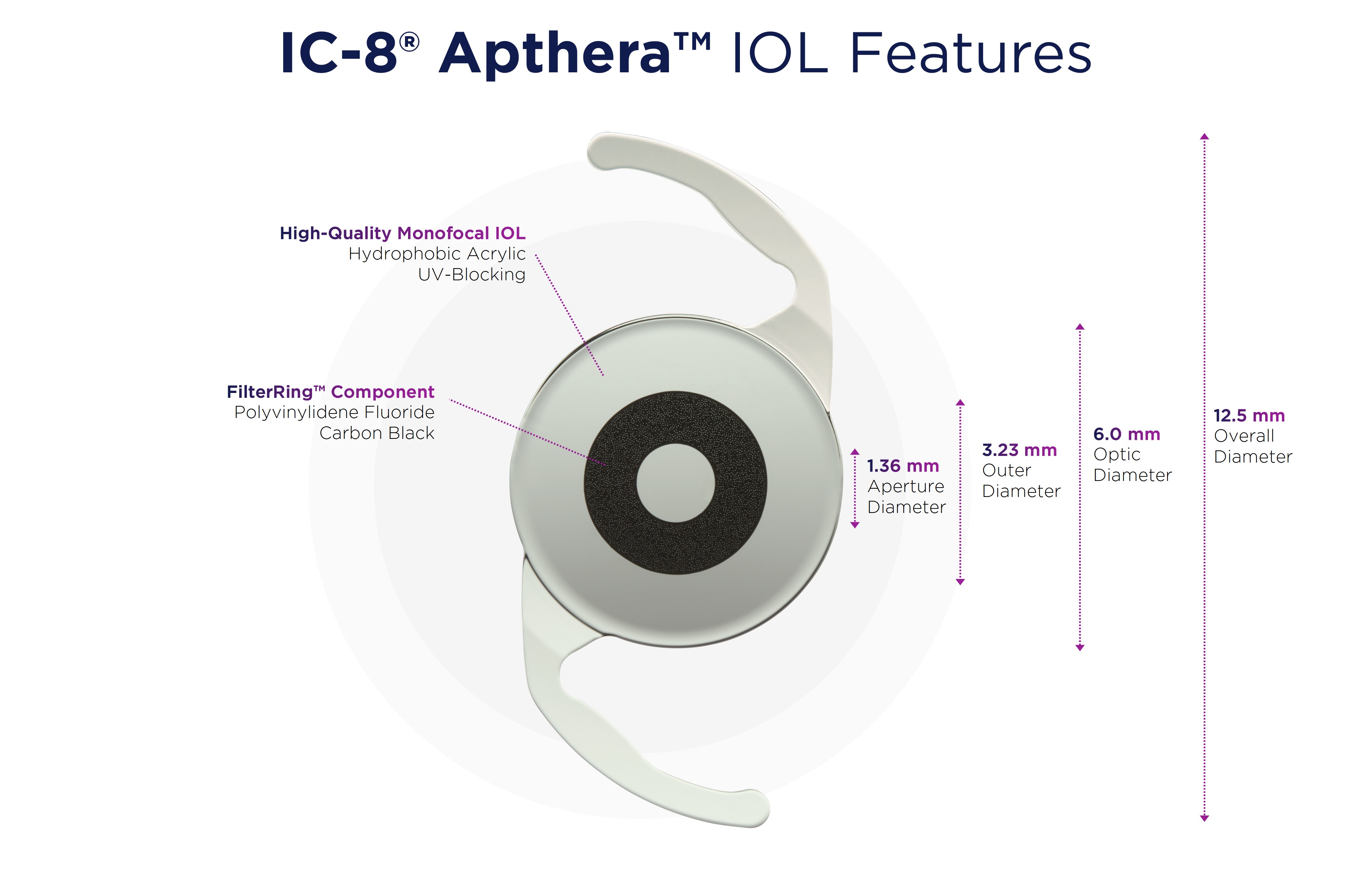
The Apthera IOL, with its proprietary small aperture technology, provides excellent distance vision as well as clear intermediate and near vision, effectively mitigating the effects of presbyopia, according ot AcuFocus. This is unlike other available presbyopia-correcting lens designs, which have complex optics that split, shift, or stretch light to provide clear vision at more than one discrete focal point.
“I am thrilled to have the privilege of treating the first commercial patients with the Apthera IOL and its novel mechanism of action,” said Dr. Thompson. “Featuring its embedded FilterRing component, this unique lens mitigates presbyopia’s effects in an elegantly simple way—by filtering out peripheral defocused and aberrated light that degrades image quality to allow only central focused light to be delivered to the retina. Now, even our patients with as much as 1.5 D of corneal astigmatism can enjoy a continuous range of vision from far through intermediate and near.”
FDA approval of the Apthera IOL is based on data from the U.S. Investigational Device Exemption study that evaluated the safety and effectiveness of the Apthera IOL implanted in one eye and a monofocal or monofocal toric IOL implanted in the fellow eye. A total of 453 subjects were enrolled and followed for 12 months. Outcomes for the Apthera IOL group (n=343) were compared to a control group (n=110) receiving a monofocal or monofocal toric IOL in both eyes. Apthera IOL treated eyes maintained 2 D of extended depth of focus and demonstrated 0.91 D of additional distance-corrected range of vision benefit over monofocal IOL eyes at 0.2 logMAR threshold, exceeding the 0.50 D ANSI criterion for extended depth of focus IOLs. Apthera IOL subjects achieved equivalent uncorrected distance vision and statistically superior intermediate and near vision compared to control subjects. Apthera IOL subjects also achieved comparable binocular contrast sensitivity to control subjects in both photopic and mesopic conditions, a first reported for an extended depth of focus lens.
Over the coming weeks, AcuFocus will be expanding its team and training surgeons in advance of the expected full US launch of the Apthera IOL during the second quarter of this year.
